 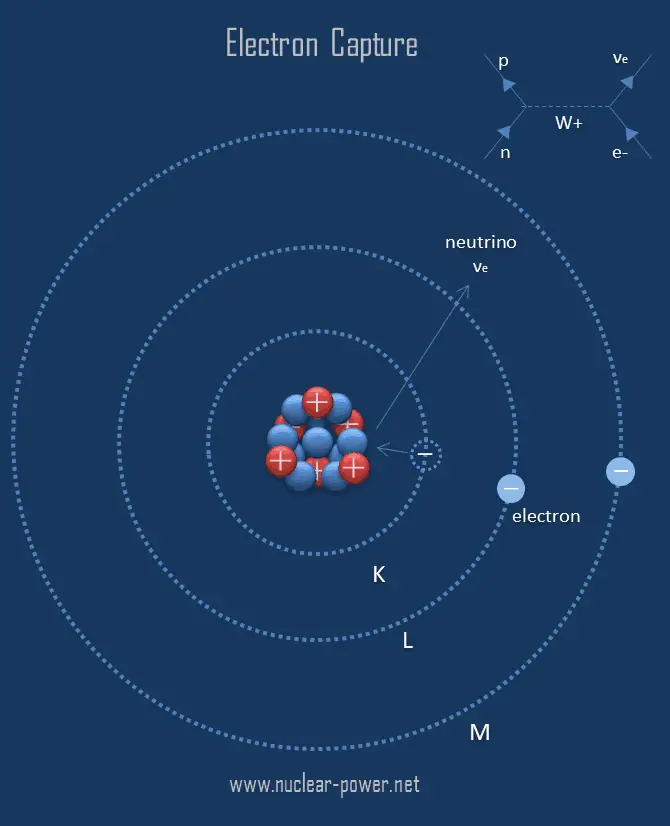
In this process, a positron will be emitted from the nucleus. The device was invented in 1957 by James Lovelock 1 2 3 4 and is used in gas chromatography to detect trace amounts of chemical compounds in a sample. In positron decay, the mass number of an atom doesn’t change, but the atomic number decreases by one. An electron capture detector ( ECD) is a device for detecting atoms and molecules in a gas through the attachment of electrons via electron capture ionization. The energy given off in this reaction is carried by an x-ray photon, which is. But potassium (K) is converted into argon (Ar) as the atomic number changes by one unit.Įquation electron capture reaction is similar to that of positron decay. The temperature dependence of the electron capture process was investigated for elucidating high electron capture sensitivity of the Cr(III) complexes of. Electron capture leads to a decrease of one in the charge on the nucleus. The equation for electron capture can be written as, Electron capture reactions are also called K-electron capture or L-electron capture, because electrons are absorbed from the K or L shell of the atom. The energy released during this process comes out as gamma rays. 
Then the absorbed electron and one proton combines to form a neutron, thereby balancing the number of protons and neutrons in the nucleus. The nucleus absorbs an electron from the K-shell or L-shell of the atom. The three processes are electron emission, positron (positive electron) emission, and electron capture. A nucleus undergoes electron capture, if it contains more number of protons compared to that of neutrons. beta decay, any of three processes of radioactive disintegration by which some unstable atomic nuclei spontaneously dissipate excess energy and undergo a change of one unit of positive charge without any change in mass number. /electron-capture-4581dd61542742dc9a86000e0af3ac2f.jpg)
This process involves emission of high energy gamma rays from the nucleus.Įlectron capture is a nuclear process. Electron Capture An alternate way for a nuclide to increase its neutron to proton ratio is by a phenomenon called electron capture. for the + (positron) emission form a nucleus, there is another. A nucleus rich in protons absorbs an inner electron from the atom. Related Videos Electron capture occurs more often than positron emission in heavy elements. Hint: Electron capture is a nuclear process. A gas chromatography/electron capture/negative chemical ionization high-resolution mass spectrometry (GC/EC/NCI-HRMS) method was developed for quantitating. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
